 
Sharing of energy can be related to measurable thermodynamic properties of substances – that is, of reactants and products. Now we need to understand how the direction and extent of the spreading and We have explored how the tendency of thermal energy to disperse as widely as possible is what drives all spontaneous processes, including chemical reactions. Rudolf Clausius originated the concept as energy gone to waste in the early 1850s, and its definition went through a number of more precise definitions over the next 15 years. It is also widely misrepresented as a measure of disorder, as we discuss below. Absolute entropies of most common substances are tabulated, allowing us to calculate the entropy of a reaction in the same way we can calculate enthalpy of reaction from standard enthalpies of formation.Įntropy is one of the most fundamental concepts of physical science, with far-reaching consequences ranging from cosmology to chemistry. 
The second section discusses the meaning of entropy, and what disorder means on a microscopic level.Įntropy is a state function, which means we can apply Hess' Law to it. A reversible process can be modeled as a series of tiny steps, while an irreversible process must be modeled as a single large change. The first section explains the difference between reversible and irreversible processes. Because of this, the second law of thermodynamics explains why a perpetual motion machine can never exist. This is because some of the energy from your car engine is lost as heat. If you touch the hood of your car while the engine is running, the hood of the car will feel hot. One consequence of the second law of thermodynamics is that in any engine there will be some energy lost as heat that cannot be harnessed to do work. The second law of thermodynamics states that the entropy of the universe is always increasing. Entropy is a measure of the disorder of a system, measured in joules (J). D S o fcan beĬalculated for chemical compounds using the S o values of elements from which the compound is formed.The second law of thermodynamics involves a thermodynamic quantity we call entropy (S). Of 1 mole of compound from the elements present in the standard conditions. Standard entropy change of formation, D S o f is defined as the entropy of formation This D S o is the standard entropy change of the reaction. When the standard entropies, S o of various substancesĬhange of a chemical process or reaction is written as 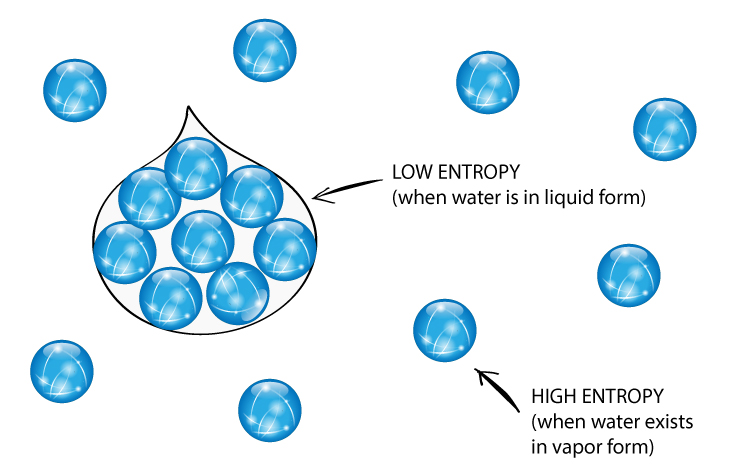
Or compounds at any temperature above 0 o K always have positive values. The standard entropies of all substances either elements The absolute entropy of a pure substance at 25 o C (298 K) Of the process involving D H, amount of enthalpy change, at constant pressure. D S is also calculated from D H as D S = D H / T where T is the temperature Since D H is the heat absorbed (or) evolved in the process at constant T and pressure P. Viii) Entropy change is related to enthalpy change as cgs units of entropy is cal.K -1 denoted as eu. Since entropy also depends on the quantity of the substance, unit of entropy is calories perĭegree per mole (or) eu. Units of entropy: The dimension of entropy are energy in terms of heat X temperature -1. Process, at constant T, D S is positive ( D S > 0).ĭ S is positive ( D S > 0). Universe remains constant although the entropy of the universe tends to a maximum. The entropy of the universe increases in an irreversible ForĮntropy change of the universe during a reversible process is Zero. Undergoes a physical (or) a chemical process, there occurs a change in the entropy of the system and also in itsĮntropy of the system and its surroundings is termed as the entropy change of the universe brought about by the process. Hence entropy may be regarded as a measure of disorder (or) randomness of the molecules of the system. Process is accompanied by increase in the 'disorder' (or) 'randomness' of the molecules constituting the system.Įntropy increases in all spontaneous processes. Process D q = (q 2-q 1) divided by temperature (T) of the process is a state Heat, q is not a state function, But for a reversible Process at constant temperature to the temperature of the system under process. Entropy change ' D S' of a system under a process is defined as the constantĮqual to the ratio of the heat change accompanying a The term 'S' entropy is evolved from theįormulation of II law of thermodynamics as a 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
